AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Anode and cathode definition8/18/2023  
Traditionally, the electrodes have been located at the battery’s ends. The positive electrode is another name for it.Īnode, cathode, and electrolyte are the three essential components of a simple battery. The anode is the electrode where the cell’s current leaves and oxidation occurs. In a galvanic or voltaic cell, an anode becomes a cathode (negative charge).Ī cathode becomes an anode in a galvanic or voltaic cell (positive charge). In electrolytes cells, it has an overabundance of negative charges. In electrolytes cells, it has an overabundance of positive charges. What kind of reaction takes place at the cathode and anode?Īn oxidation process occurs at the anode in both galvanic and electrolytic cells.Ī reduction reaction occurs at the cathode in both galvanic and electrolytic cells. The cathode is the electrode in the battery with the negative (-) terminal.Īn anode (positive charge) is sometimes called an electron donor.Īn electron acceptor is a cathode (negative charge). The anode is the electrode with the positive (+) terminal showing in the battery. The cathode is the electrode that conducts electric current from the battery’s negative terminal to the positive terminal. What are the anode and cathode?Īn anode is an electrode that conducts conventional current from the battery’s positive terminal to the negative terminal. When the electrode gains electrons released by the electrolyte and experiences reduction, the electrode performs the opposite operation. Oxidation occurs when an electrode loses electrons and is received by the electrolyte. 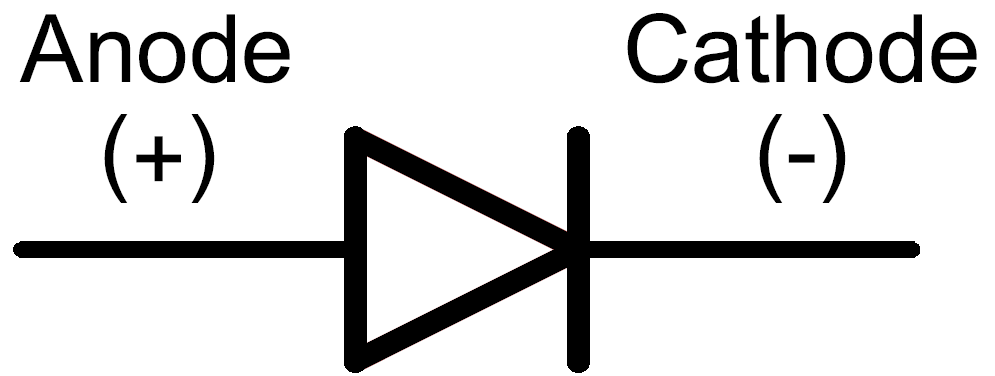
It describes the conductive phase in which charged carriers are transferred. Within the electrochemical cell, an electrode is an electrical conductor/semiconductor. It is defined as a surface on which a redox reaction occurs between the metal and the solution. The current enters and exits the electrolyte via the electrode, which acts as a metallic contact. Anodes and cathodes are used in a variety of other applications.Īn electrode is a critical component of an electrochemical cell that comes into contact with electrolytes.Main Differences Between Cathode and Anode.Why is it important to understand the difference between an anode and a cathode in a battery?. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
